Your Location:Home >Products >Organic phosphines >Phenyl phosphines >855-38-9
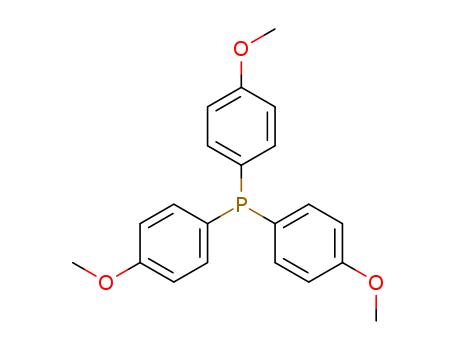

Product Details
Chemical Properties
White to pale yellow crystalline powder
Uses
suzuki reaction
Uses
Tris(4-methoxyphenyl)phosphine is a useful intermediate employed in organic synthesis and in other chemical processes.
InChI:InChI=1/C21H21O3P/c1-22-16-4-10-19(11-5-16)25(20-12-6-17(23-2)7-13-20)21-14-8-18(24-3)9-15-21/h4-15H,1-3H3
Chlorobenzenes are important starting materials for the preparation of commercially valuable triarylphosphines and tetraarylphosphonium salts, but their use for the direct arylation of elemental phosphorus has been elusive. Here we describe a simple photochemical route toward such products. UV-LED irradiation (365 nm) of chlorobenzenes, white phosphorus (P4) and the organic superphotoreductant tetrakis(dimethylamino)ethylene (TDAE) affords the desired arylphosphorus compounds in a single reaction step.
Reduction of phosphine oxides into the corresponding phosphines using PhSiH3 as a reducing agent and Ph3C+[B(C6F5)4]? as an initiator is described. The process is highly efficient, reducing a broad range of secondary and tertiary alkyl and arylphosphines, bearing various functional groups in generally good yields. The reaction is believed to proceed through the generation of a silyl cation, which reaction with the phosphine oxide provides a phosphonium salt, further reduced by the silane to afford the desired phosphine along with siloxanes. (Figure presented.).
The invention aims to provide an aryl phosphine oxide compound as a raw material, wherein P=O keys are activated by an acid anhydride and alkali is continued. The preparation of the phosphine (III) compound is carried out under the action of a crown ether and a reducing agent. The method has the advantages of cheap and easily available raw materials, simple operation, high atomic economy and the like. Compared with a traditional reduction mode, the method is ingenious in design, waste emission is reduced, separation of intermediate products is omitted, and related reagents such as silicon hydrogen, aluminum, boron and the like with higher price can be avoided. And the reaction suitability is extensive.
Detailed 31P{1H} NMR spectroscopic investigations provide deeper insight into the complex, multi-step mechanisms involved in the recently reported photocatalytic arylation of white phosphorus (P4). Specifically, these studies have identified a number of previously unrecognized side products, which arise from an unexpected non-innocent behavior of the commonly employed terminal reductant Et3N. The different rate of formation of these products explains discrepancies in the performance of the two most effective catalysts, [Ir(dtbbpy)(ppy)2][PF6] (dtbbpy=4,4′-di-tert-butyl-2,2′-bipyridine) and 3DPAFIPN. Inspired by the observation of PH3 as a minor intermediate, we have developed the first catalytic procedure for the arylation of this key industrial compound. Similar to P4 arylation, this method affords valuable triarylphosphines or tetraarylphosphonium salts depending on the steric profile of the aryl substituents.
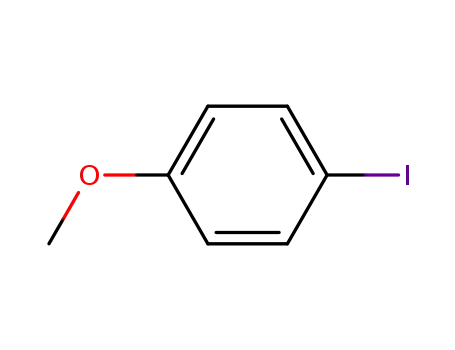
para-iodoanisole

P(p-CH3OC6H4)3

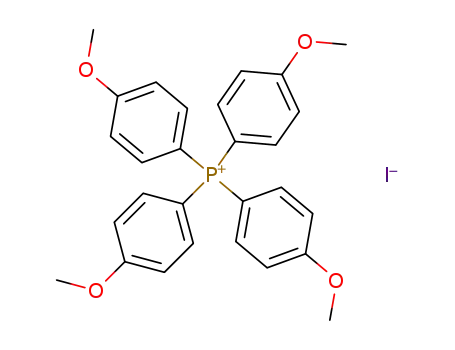
tetrakis(4-methoxyphenyl)phosphonium iodide
| Conditions | Yield |
|---|---|
|
With
dihydrogen phosphide sodium; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; triethylamine;
In
acetonitrile;
at 20 ℃;
for 24h;
Reagent/catalyst;
Sealed tube;
Irradiation;
Schlenk technique;
Glovebox;
Inert atmosphere;
|
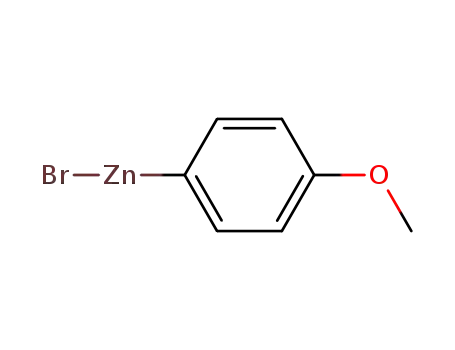
(4-methoxyphenyl)zinc(II) bromide

P(p-CH3OC6H4)3
| Conditions | Yield |
|---|---|
|
With
phosphorus trichloride;
In
acetonitrile;
at 20 ℃;
for 2h;
|
45% |
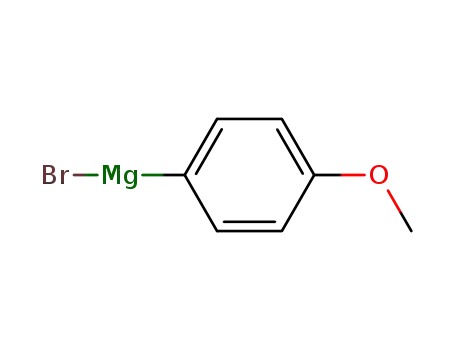
4-methoxyphenyl magnesium bromide
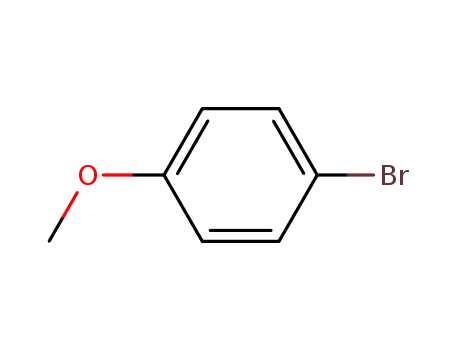
1-bromo-4-methoxy-benzene
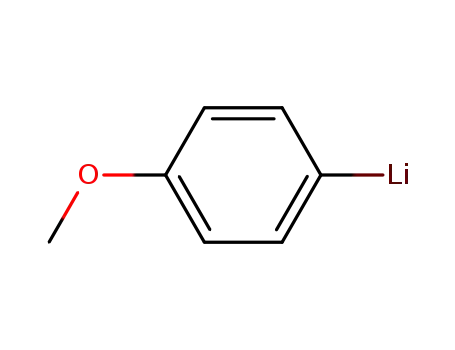
p-methoxyphenyllithium

(4-methoxyphenyl)zinc(II) bromide
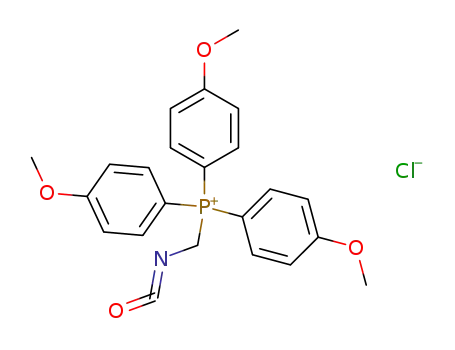
Isocyanatomethyl-tris-(4-methoxy-phenyl)-phosphonium; chloride
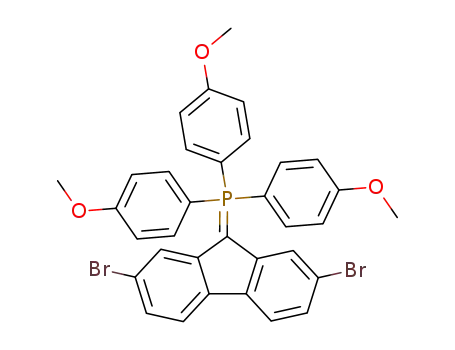
Tris-(4-methoxy-phenyl)-<2,7-dibrom-fluorenyliden-(9)>-phosphoran
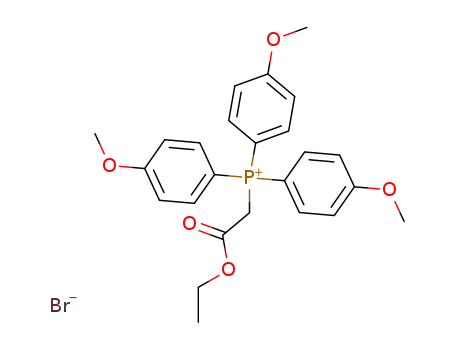
Tris-(4-methoxy-phenyl)-ethoxycarbonyl-methylen-phosphoran-hydrobromid
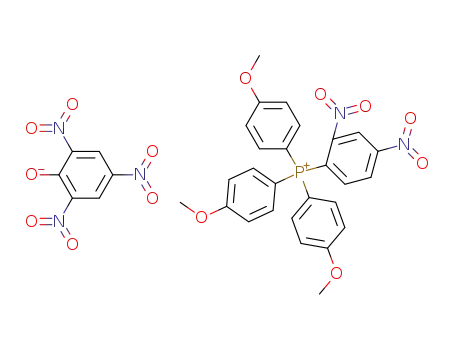
Tris-<4'-methoxy-phenyl>-<2,4-dinitro-phenyl>-phosphonium-pikrat
CAS:189367-54-2
CAS:240417-00-9
CAS:6224-63-1
CAS:13885-09-1