Your Location:Home >Products >Functional intermediates >538-58-9
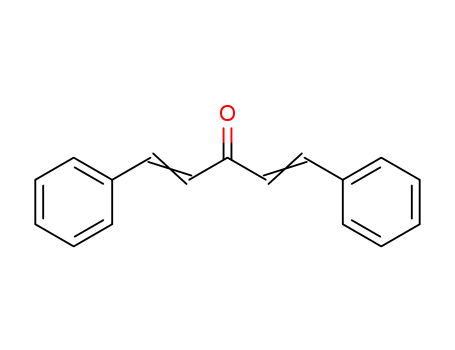

Product Details
Chemical Properties
trans,trans-Dibenzylideneacetone is a crystalline solid, melting point 110-111℃; cis-trans is a light yellow needle crystal, melting point 60℃; cis-cis is a yellow oily liquid, boiling point 130℃ (2.7Pa). Soluble in ethanol, acetone, chloroform, insoluble in water.
Uses
Dibenzylideneacetone is used as a ligand to prepare palladium catalysts, which are widely used in catalytic hydrogenation, coupling, carbonylation, alkyne ring trimerization, etc.
Preparation
Dibenzylideneacetone is obtained by the reaction of benzaldehyde and acetone. The reaction was carried out in aqueous ethanol solution at a reaction temperature of 20-25°C with a yield of 78%.
Synthesis Reference(s)
Chemistry Letters, 9, p. 51, 1980The Journal of Organic Chemistry, 45, p. 3840, 1980 DOI: 10.1021/jo01307a022Organic Syntheses, Coll. Vol. 2, p. 167, 1943
Purification Methods
Crystallise the ketone from hot ethyl acetate (2.5mL/g) or EtOH. [Beilstein 7 IV 1747.]
InChI:InChI=1/C17H14O/c18-17(13-11-15-7-3-1-4-8-15)14-12-16-9-5-2-6-10-16/h1-14H/b13-11+,14-12u
The aim of this work was to prepare and characterize inclusion complexes between a synthetic curcumin analog (dibenzalacetone, DBA) and beta-cyclodextrin (β-CD); and to evaluate their in vitro antileishmanial activity. DBA was synthetized and characterized by spectroscopic methods and the inclusion complexes were obtained by kneading and lyophilization (LIO) in 1:1 and 1:2 stoichiometries. Phase solubility and dissolution assays showed a 40-fold increase in the aqueous solubility of DBA and its complete dissolution from LIO 1:1 formulation after 120 min respectively. Solid-state characterization by differential scanning calorimetry and near infrared spectroscopy demonstrated the inclusion of DBA in the β-CD cavity at the molar ratios tested, with LIO 1:1 formulation being the most stable. Using nuclear magnetic resonance experiments, the protons inside the cavity of β-CD were the most affected after the inclusion of DBA molecule. The cellular viability of THP-1 macrophage cells treated with plain DBA, β-CD and DBA/CD inclusion complexes showed that the plain DBA and DBA/CD at 1:2 stoichiometry presented toxicity, while β-CD alone and DBA/CD at 1:1 stoichiometry showed no toxicity up to 640 μg mL?1. The in vitro assay with free-living promastigotes demonstrated that plain DBA and β-CD had IC50 of 320 μg mL?1 respectively, while only inclusion complexes with 1:1 stoichiometry showed antiproliferative activity with IC50 = 51.3 μg mL?1. Using the amastigote intracellular forms, there was also a difference between the plain and β-CD complexed DBA with complexes of 1:1 and 1:2 stoichiometry presenting EC50 = 66.3 μg mL?1 and 58.9 μg mL?1 respectively. The study concluded that DBA/CD at 1:1 molar ratio has the potential to decrease the intrinsic toxicity of plain DBA towards Leishmania host cells, which may be a therapeutic advantage in the application of these compounds.
Herein, a wet chemical epitaxial growth method was employed to fabricate a cactus-like CuFeO2/ZnO heterojunction for the photocatalytic reduction of benzaldehyde to benzyl alcohol. The 1D ZnO nanorods in the heterojunction make contact with the 2D CuFeO2 nanoflakes at the atomic level, therefore providing a fast charge transfer channel along the direction parallel to the CuFeO2c-axis, leading to efficient charge separation and improved photocatalytic performance.
-
1,5-Diphenylpenta-1,4-dien-3-one ( dibenzalacetone, DBA) was synthesized by a base-catalyzed aldol condensation reaction between benzaldehyde and acetone. High quality single crystals have been grown by the slow evaporation of ethanol solution and the crystal belongs to monoclinic system with centrosymmetric space group C 2/c. The DBA crystals are transparent in the entire visible region and have a lower optical cutoff at ~440 nm. It is stable up to 119 °C and has a good chemical stability. The high resolution X-ray diffraction curve (DC) indicates that the specimen is free from structural grain boundaries. Molecular packing leads to a centrosymmetric arrangement resulting in zero second harmonic generation (SHG; X(2)=0) efficiency.
The interaction of Dibenzalacetone (DBA) with β-Cyclodextrin (β-CD) producing an inclusion complex was carried out by co-precipitation method. The binding constant was determined using steady state and time-resolved fluorescence spectroscopy and the results suggested that the inclusion complex preferred 1:1 stoichiometry. The complex was characterized by UV-Visible, infrared spectroscopy (FT-IR), differential scanning calorimetry (DSC) and X-ray diffractometry (XRD). The morphological characteristics of the solid complex were analyzed by Scanning electron microscope (SEM) and Atomic force microscope (AFM). The structure of 1:1 inclusion complex of DBA with β-CD is proposed. The Docking study reveals that this structure was found to be highly probable and energitically favorable model.
A novel, heterogeneous and homogeneous basic nanocatalysts were synthesized by grafting of lithium and cesium on zagrosian natural asphalt sulfonate (Li/Cs-Zagronas). The activity of these catalysts was examined in the Claisen–Schmidt and Knoevenagel condensations under mild reaction conditions. Li/Cs-Zagronas were characterized by FT-IR spectroscopy, scanning electron microscopy, X-ray diffraction, energy-dispersive spectroscopy, inductively coupled plasma and thermogravimetric analysis techniques. These nanocatalysts were removed by simple filtration and reused several times without any deterioration of activity.
The synthesized 1,5 diarylpenta-1,4-dien-3-one derivatives (compounds 1-6) as synthetic curcumin analogues were tested for their potential anticancer activity against human ovarian and lung adenocarcinoma cells. The absorption, distribution, metabolism, excretion, and toxicity (ADMET/pharmacokinetic) parameters of all the compounds were predicted by admetSAR software. The pharmacokinetics, pharmacodynamics and bioactivity scores properties based on Lipinski rule and Ghose filter, calculated with the help of Molinspiration and ChemDraw. Molecular docking evaluation of all the compounds was also performed by using AutoDock Vina and iGEMDOCK against three most common human anticancer targets; epidermal growth factor receptor (EGFR), heat shock protein (Hsp 90-α), and vascular endothelial growth factor receptor-2 (VEGFR2). The obtained results were compared with the reference compound 7 and drugs 8-10 (7: GO-035; 8: Quinazolin; 9: Naquotinib and 10: Ribofuranuronamide). Finding indicates, all the compounds were potentially interacting with VEGFR2 through the average –9.1 binding energy (BE) with closer contact 1H-NMR). In vitro anti-proliferative activity was tested via MTT method against human ovarian carcinoma (PA-1) and human lung adenocarcinoma (A549) cells and further screened for apoptotic parameters such as nuclear fragmentation and ROS generation. Compound 4 exhibits good dose-dependent anti-proliferative activity (IC50 73 and 79.7 μM) against human ovarian carcinoma and human lung adenocarcinoma, respectively. Communicated by Ramaswamy H. Sarma.
Schiff bases have continued to gain attention as essential building blocks and versatile pharmacophores in drug development and drug-like molecular entities. Thus, the synthesis of Schiff bases was achieved herein via facile acetic acid catalyzed synthetic transformation of chalcones. The targeted Schiff bases and related compounds 2a-m were accessed by the treatment of amines with chalcone 1 which was previously derived through Claisen-Schmidt reaction between benzaldehyde and acetone, at ambient temperature. Structural characterization was achieved via physicochemical properties and the use of IR, UV, 1H and 13C NMR which were spectroscopic techniques. The compounds have essential candidature for further study, in biological activity so as to unleash their medicinal potential.
In this work we report the synthesis of two fluorescent 2-pyrazoline derivatives exhibiting remarkable (fluoro)solvatochromic behavior. The pyrazolines were synthesized in one step from the corresponding monocarbonyl curcuminoids and 4-hydrazinobenzoic acid in high yield and purity and were fully characterized by means of NMR and FT-IR spectroscopy and HRMS spectrometry. The structure of the novel derivative 2 was also characterized by means of X-ray crystallography. Both compounds were studied computationally in the gas phase. Additionally, the effects of solvent polarity on the absorption and fluorescence spectra of these derivatives were investigated in a solvent group consisting of various neat molecular solvents exhibiting hydrogen bond donating (HBD), hydrogen bond accepting (HBA), and/or dipolar behavior. The solvent effects observed were quantified and rationalized by employing suitable multiparametric Linear Solvation Energy Relationships (LSERs) involving dipolarity, HBD-acidity and HBA-basicity terms. The contribution of each of these parameters provided insights on the predominant solute-solvent interactions occurring in solution. Importantly, some differences in the (fluoro)solvatochromic aptitude/behavior were revealed among the derivatives and they were rationalized on the basis of their structural diversity.
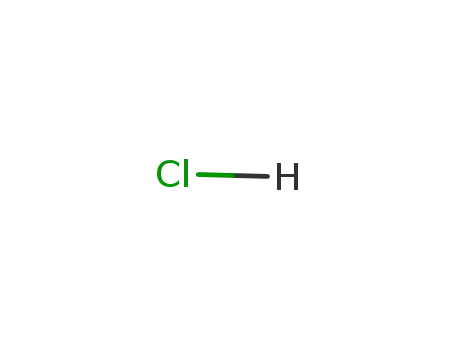
hydrogenchloride

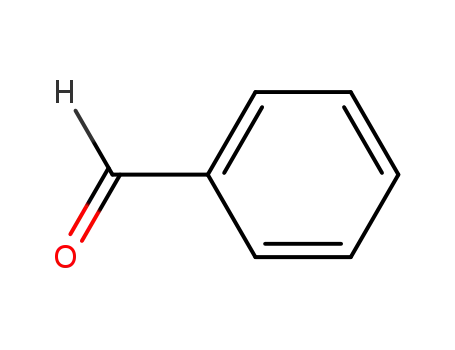
benzaldehyde

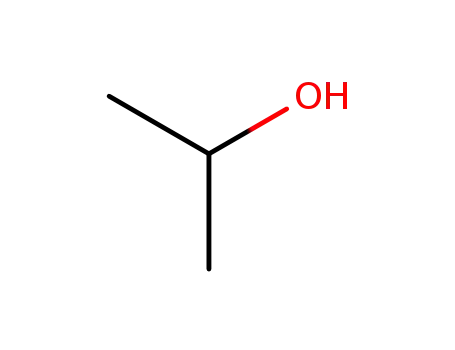
isopropyl alcohol

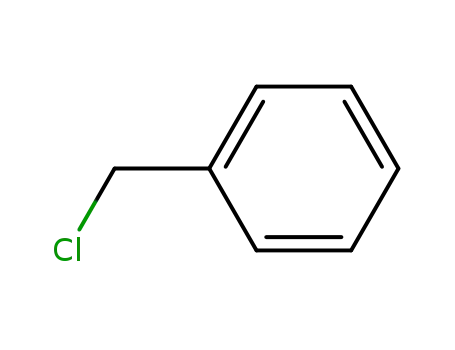
benzyl chloride

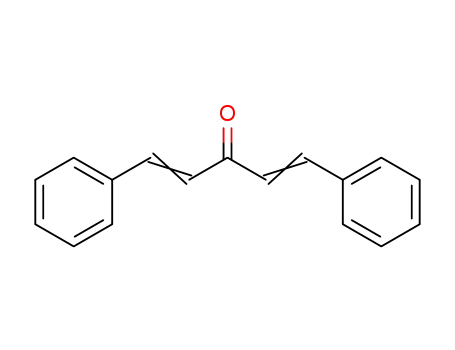
1,5-diphenyl-1,4-pentadiene-3-one
| Conditions | Yield |
|---|---|
|
|

benzaldehyde


1,5-diphenyl-1,4-pentadiene-3-one

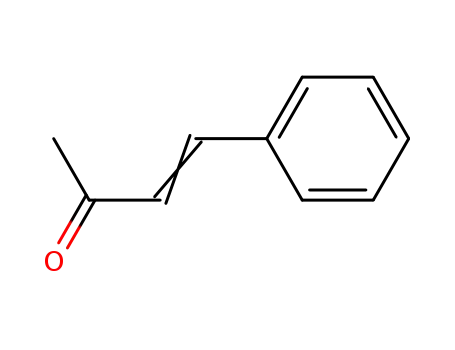
1-Phenylbut-1-en-3-one

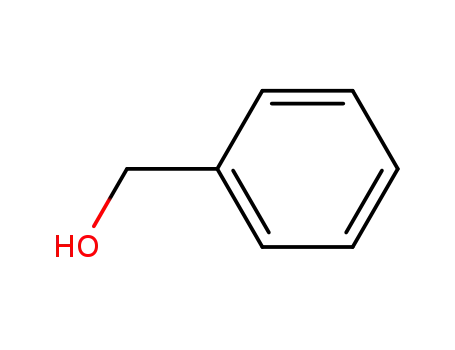
benzyl alcohol
| Conditions | Yield |
|---|---|
|
gadolinium(III) isopropoxide;
In
benzene;
at 0 ℃;
for 2h;
|
2.5% 0.6% 52% |
|
With
sodium 2,6-di-tert-butyl-4-methoxyphenolate; C23H21FeN5O2P(1+)*BF4(1-); isopropyl alcohol;
In
tetrahydrofuran;
at 60 ℃;
for 20h;
Reagent/catalyst;
Temperature;
Catalytic behavior;
|
53 %Spectr. 22 %Spectr. 6 %Spectr. |
|
With
potassium tert-butylate; C23H21FeN5O2P(1+)*BF4(1-); isopropyl alcohol;
In
tetrahydrofuran;
at 40 ℃;
for 20h;
Catalytic behavior;
|
17 %Spectr. 11 %Spectr. 9 %Spectr. |
|
With
C23H21FeN5O2P(1+)*BF4(1-); isopropyl alcohol; potassium hydroxide;
In
tetrahydrofuran;
at 80 ℃;
for 20h;
Catalytic behavior;
|
35 %Spectr. 11 %Spectr. 7 %Spectr. |
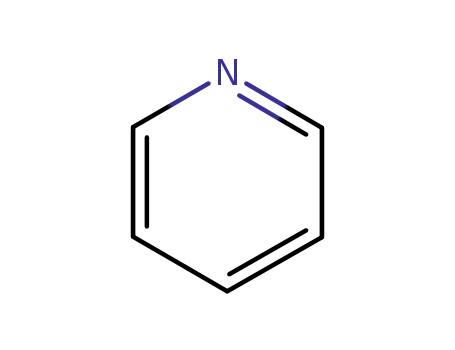
pyridine
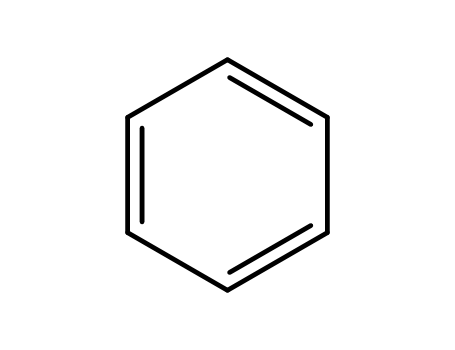
benzene
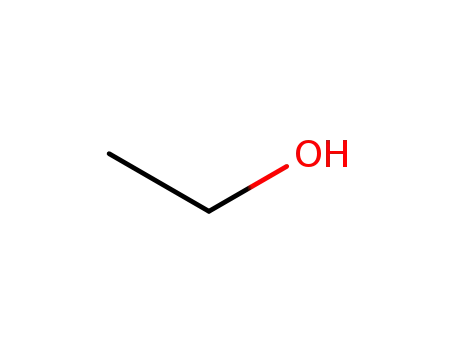
ethanol
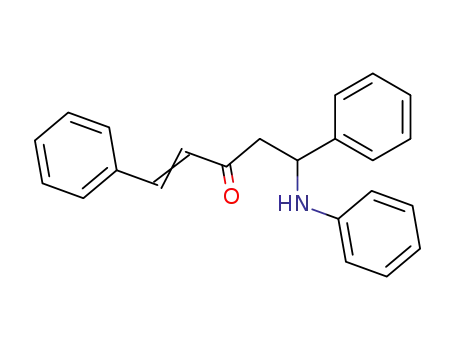
5-anilino-1,5-diphenyl-pent-1-en-3-one
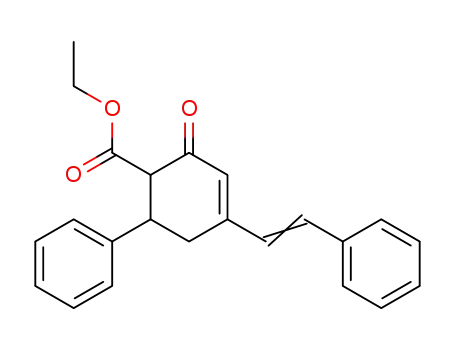
ethyl 5-phenyl-3-styryl-2-cyclohexen-1-one-6-carboxylate
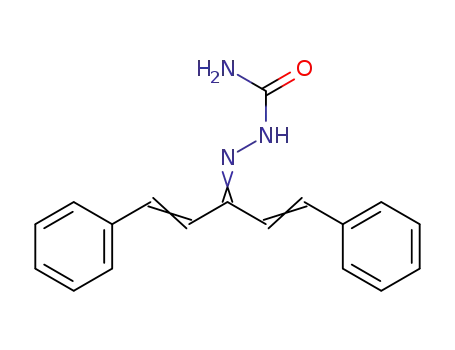
1,5-diphenyl-penta-1,4-dien-3-one semicarbazone
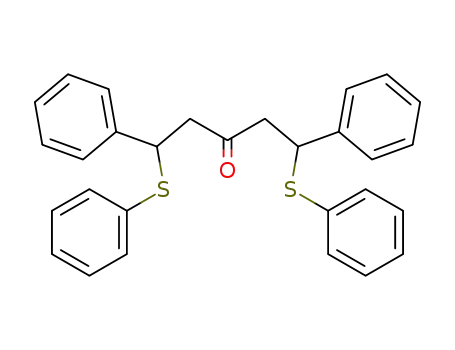
1,5-diphenyl-1,5-bis(phenylsulfenyl)pentan-3-one
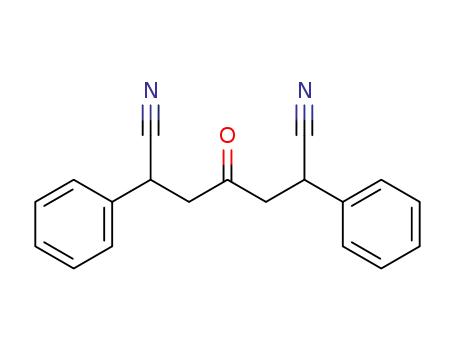
4-oxo-2,6-diphenyl-heptanedinitrile
CAS:932710-63-9
Molecular Formula:C16H28NP
Molecular Weight:265.37
CAS:860465-14-1