Your Location:Home >Products >Functional intermediates >3425-89-6

Product Details
|
Flammability and Explosibility |
Nonflammable |
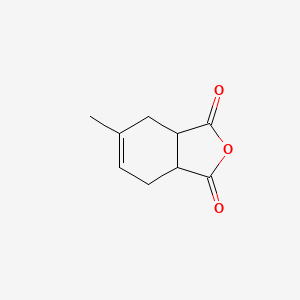
InChI:InChI=1/C9H10O3/c1-5-2-3-6-7(4-5)9(11)12-8(6)10/h2-3,5-7H,4H2,1H3
Polarization transfer is demonstrated as...
The kinetics of the reaction of maleic a...
Cyclic imides belong to a well-known cla...
A high-pressure FTIR spectroscopy was us...
-
A range of Lewis acidic metal chlorides ...
An efficient dehydrogenative Diels-Alder...
maleic anhydride

isoprene

4-methyl-1,2,3,6-tetrahydrophthalic anhydride
| Conditions | Yield |
|---|---|
|
for 0.0833333h; stereoselective reaction; Microwave irradiation; Sealed tube;
|
99% |
|
With methyltrioxorhenium(VII); In acetone; for 0.333333h; Ambient temperature;
|
95% |
|
With silica-supported ionic liquid [1-methyl-3-(triethoxysilylpropyl)imidazolium ][Ga3Cl10]; at 25 ℃; for 3h; Ionic liquid;
|
93% |
|
In toluene; at 155 ℃; for 0.0833333h; Microwave irradiation;
|
92% |
|
In o-xylene; at 25 ℃; for 72h; Sealed tube; Inert atmosphere; Sonication;
|
65% |
|
aluminium trichloride; In carbon dioxide; benzene; at 32.9 ℃; under 55879.4 Torr; Mechanism; supercritical conditions; 0 -> 1.0 min; other pressure: 7.85 MPa
|
|
|
|
|
|
aluminium trichloride; In carbon dioxide; benzene; at 32.9 ℃; for 0.0166667h; under 55879.4 Torr; supercritical conditions;
|
|
|
In 1,4-dioxane; at 30 ℃; Yield given;
|
|
|
In 1,4-dioxane; at 30 ℃; Rate constant; Kinetics;
|
|
|
In xylene; at 25 ℃; kinetic isotope effects;
|
|
|
|
|
|
In nitrobenzene; at 28 ℃; Kinetics;
|
|
|
In carbon dioxide; at 60 ℃; under 76000 Torr; Kinetics;
|
|
|
In neat (no solvent); at 30 ℃; for 1h; Green chemistry;
|
93.4 %Chromat. |
|
With N-[2-(D-glucopyranosyl)ethyl]-N,N,N-trimethylammonium bistriflamide; at 20 ℃; for 1h; diastereoselective reaction; Inert atmosphere; Green chemistry;
|
99 %Chromat. |
|
In tetrahydrofuran; at 20 ℃; for 2h;
|
|
|
In dichloromethane; at 20 ℃; for 5h; diastereoselective reaction;
|
isoprene

4-methyl-1,2,3,6-tetrahydrophthalic anhydride
| Conditions | Yield |
|---|---|
|
|
|
|
|
(+/-)-4-methyl-cyclohex-4-ene-1r,2t-dicarbonitrile
maleic anhydride
isoprene
cis-3a,4,7,7a-tetrahydro-5-methyl-1,3-isobenzofurandione
(+/-)-4c-methyl-cyclohexane-1r,2t-dicarboxylic acid-anhydride
(+/-)-4t-methyl-cyclohexane-1r,2t-dicarboxylic acid-anhydride
(+/-)-4t-methyl-cyclohexane-1r,2c-dicarboxylic acid-anhydride
CAS:120-74-1
Molecular Formula:C<sub>8</sub>H<sub>10</sub>O<sub>2</sub>
Molecular Weight:138.16
CAS:935-79-5
Molecular Formula:C<sub>8</sub>H<sub>8</sub> O<sub>3</sub>
Molecular Weight:152.15
CAS:1450933-18-2
Molecular Formula:C25H15Br
Molecular Weight:395.3